ISO 13485 Certification In Tanzania
Precision Healthcare Starts Here – ISO 13485 Certification Trusted Across Tanzania’s Medical Sector!
ISO 13485 Certification in Tanzania assists medical device producers and associated institutions in verifying their capability to consistently fulfill both regulatory and buyer expectations concerning the device’s quality and safety features. ISO 13485 reinforces proper process control, robust risk management, and thorough product traceability as the globally accepted standard for a quality management system for the medical device industry.
Tanzanian companies can now improve their operational efficiency, gain international trust, expand their market reach, and enhance their reputation by receiving this certification. It builds trust with patients, regulatory authorities, and other stakeholders.
Visit ISO-certification-Tanzania.com today to embark on your certification journey and access dedicated professionals who will ensure your ISO 13485 compliance and implementation is as seamless as possible.
What is ISO 13485 Certification?
Get Free Consultation:
ISO 13485 is a standard that defines the requirements of a quality management system of an organization which is involved in the design, production, servicing, and installation of medical devices.
In the simplest of terms, ISO 13485 is put in place so that medical device companies can ensure that their products are safe, reliable, and meets the expectation of their clients and the laws in place. It emphasizes the need for risk management, product traceability, as well as hygiene and documentation control during and after the manufacture of the product.
ISO 13485 is important in establishing confidence for regulators, healthcare practitioners, and patients in the confidence in the disturbing the industries in which products are important in sakes and quality are of the essence.
Importance of ISO 13485 Certification in Tanzania:
The ISO 13485 Certification in Tanzania is crucial for the development of the country’s medical device industry because it ensures products are safe, trustworthy, and compliant with quality standards. This certification fulfills Tanzania suppliers and manufacturers’ access to international markets and earns the trust of the hospitals, regulators, and medical professionals as the need for sophisticated healthcare tools escalates.
Furthermore, it enhances the opportunities with regards to the exports, collaborations, and the government tenders. ISO 13485 Certification assists in mitigating risks and sharpening the competition with regards to the organization. For companies issued with the healthcare sector issued with the healthcare sector, ISO 13485 Certification is in place to help companies achieve these goals.
How to Get ISO 13485 Certification in Tanzania ?
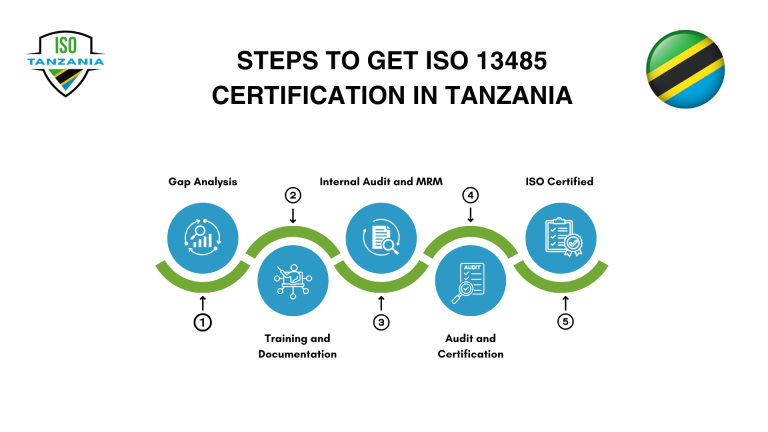
Steps to Achieve ISO 13485 Certification In Tanzania:
Understand the Standard
Familiarize your team with ISO 13485 requirements for medical device quality management systems to ensure compliance and clarity on regulatory expectations.
Conduct a Gap Analysis
Compare your current practices with ISO 13485 standards to find gaps and areas that require system or procedural improvements.
Implement Quality Management System (QMS)
Develop and align your QMS with ISO 13485, including risk controls, documentation, and traceability to support regulatory compliance.
Internal Audit and Management Review
Conduct internal audits and management reviews to verify ISO 13485 compliance before undergoing the final certification process.
Certification Audit
Work with an accredited body to complete the two-stage audit. Address any findings and earn ISO 13485 certification upon approval.
Benefits of ISO 13485 Certification In Tanzania:
- Improved Product Quality: Ensures medical devices consistently meet quality, safety, and regulatory standards, minimizing defects and recalls.
- Regulatory Compliance: Aligns your operations with both local and international healthcare regulations, making it easier to enter global markets.
- Enhanced Customer Trust: Builds credibility and confidence among patients, hospitals, and regulators through a certified, reliable quality management system.
- Operational Efficiency: Streamlines internal processes and documentation, leading to fewer errors, better resource use, and lower operational costs.
- Access to Global Markets: Facilitates international partnerships and exports by complying with widely accepted global quality benchmarks.
- Competitive Advantage: Sets your company apart in Tanzania’s growing healthcare market, attracting contracts, partnerships, and investment opportunities.
Why Should Medical Device Companies in Tanzania Prioritize ISO 13485 Certification in 2025?
Now is a crucial time for medical device manufacturers and suppliers across Tanzania—including Mwanza, Dar es Salaam, Arusha, Dodoma, and Zanzibar, to consider ISO 13485 certification. As the global healthcare industry places stronger emphasis on quality and regulatory compliance, Tanzanian businesses have a unique window of opportunity to strengthen their market position.
By becoming ISO 13485 certified, your organization can:
- Gain a Competitive Edge in the Local Market: Stand out from competitors in Tanzania’s growing medical sector by showcasing your commitment to internationally recognized quality management standards.
- Be First to Access Regional Healthcare Opportunities: Position your company at the forefront of East Africa’s expanding medical trade and healthcare initiatives by ensuring your products meet global quality expectations.
- Attract Global Partnerships and Investments: ISO 13485 certification signals professionalism and product safety—two key qualities international partners and clients demand when choosing suppliers and collaborators.
Early certification isn’t just a regulatory move it’s a strategic advantage for companies aiming to lead the medical device industry in Tanzania.
What Determines the Cost of ISO 13485 Certification in Tanzania?
In Tanzania, the cost of ISO 13485 certification varies based on the organization’s size, the intricacies of the medical device operations, and the level of compliance already in place. For reliable ISO 13485 consultants in Tanzania, ISO-certification-Tanzania.com provides comprehensive options that fit your business requirements.
These key factors influence the pricing:
- Industry Type and Organization Size
- Medical Services and Devices Range and Complexity
- Documentation and Process Readiness
- Staff Headcount, Training, and Workshops
- Internal Audits and Gap Analysis
- Fees of the certification body and surveillance audit
With concern to the overall investment evaluation, ISO-certification-Tanzania.com provides a free initial consultation. As a trusted player in ISO certification in Tanzania, they guarantee the delivery of professional, reliable service and competitive prices meeting Tanzania’s regulatory framework and international market standards.
Why Choose ISO-Certification-Tanzania.com for ISO 13485 Certification in Tanzania?
At iso-certification-tanzania.com, we specialize in providing ISO 13485 Certification services for medical device companies in Tanzania. We have skilled consultants who guarantee a smooth, efficient, and compliant certification process for both domestic and international medical device industry standards. As part of our services, we help your organization improve product quality, mitigate risks, and establish enduring trust with stakeholders.
What has made medical device companies in Tanzania trust iso-certification-tanzania.com:
- Guided assistance for ISO 13485 certification all over Tanzania.
- A simplified and efficient iso certification procedure.
- Healthcare quality improvement outcomes for Tanzania’s healthcare system.
- Affordable and transparent pricing with no hidden costs.
- Established results with local and international clientele.
For quality, compliance, and enduring success, iso-certification-tanzania.com has proven to be the reliable partner for Tanzania’s burgeoning medical device industry.
FAQ's
What is ISO 13485 certification and who needs it in Tanzania?
ISO 13485 is a quality management standard for organizations involved in the design, production, installation, or servicing of medical devices. It’s essential for manufacturers, suppliers, and related service providers in Tanzania’s healthcare sector.
How long does it take to get ISO 13485 certified in Tanzania?
The timeframe varies depending on your organization’s size and readiness. On average, it can take 3 to 6 months including documentation, audits, and implementation.
Is ISO 13485 certification required to export medical devices from Tanzania?
Yes, many international markets require ISO 13485 certification as proof of compliance with global medical device regulations and quality expectations.
Can small medical businesses in Tanzania afford ISO 13485 certification?
Absolutely. ISO-certification-Tanzania.com offers cost-effective solutions tailored for small businesses, ensuring compliance without financial strain.
